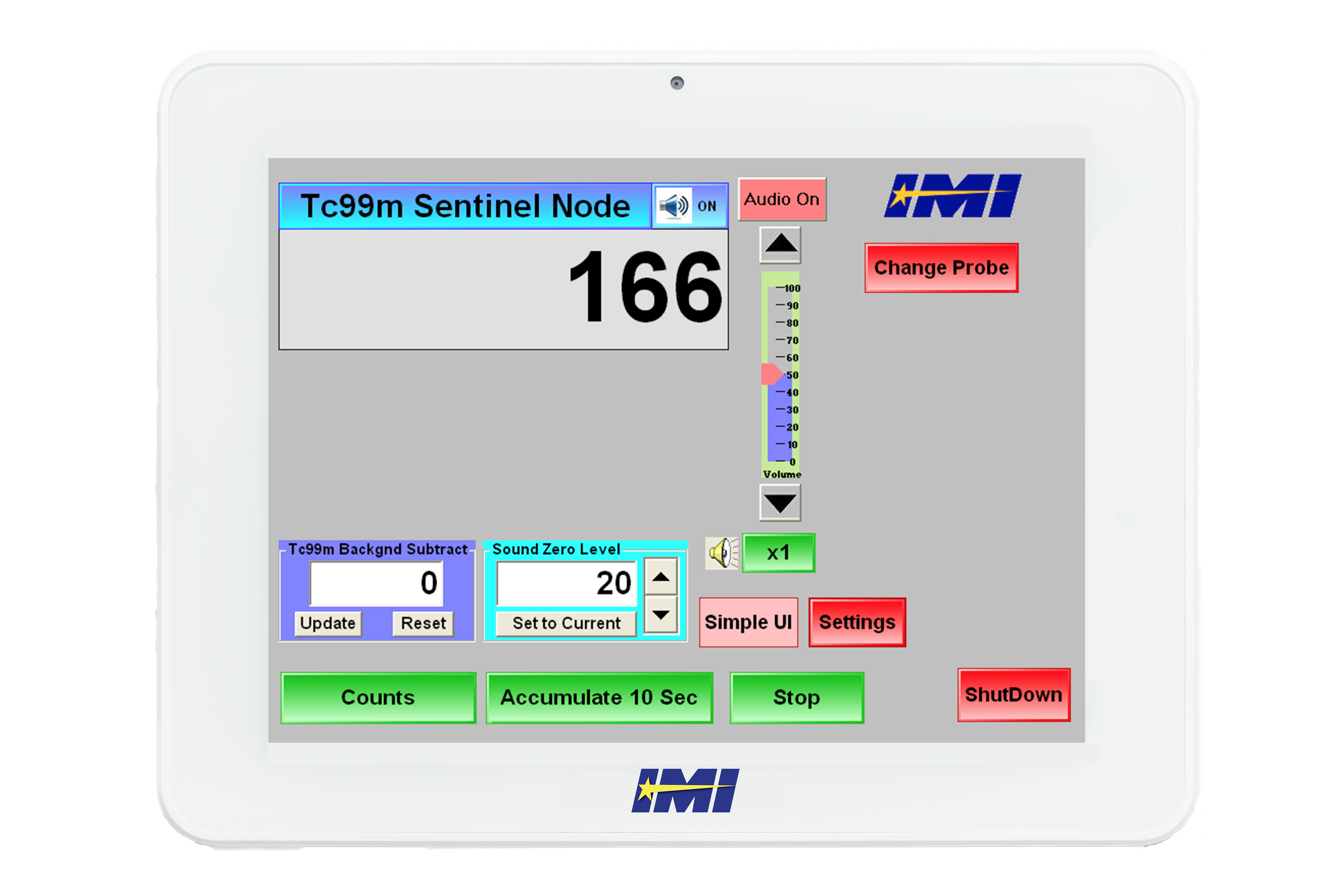
Cutting Edge Device Manufacturer Entrusts Cybernet for New Device

-
INTRAMEDICAL IMAGING, LLC.
- Industry: Healthcare
- HQ: Hawthorne, CA
 Challenge
Challenge
Over the years, Intramedical Imaging has developed several iterations of their lymph node detecting device, the Node Seeker. Initially using a large computer, then evolving to use a tablet, Intramedical was looking to develop their current model, the Node Seeker 2000, that would be powered by a tablet.
It was vital that the tablet that would serve as the brain of their device was IEC60601-1 certified safe for near-patient use in order to navigate the regulatory process involved in bringing a device to the global market. Furthermore, a long product lifecycle was key to avoid constant updates and needing to go back to the drawing board. A fanless design was another critical piece of the puzzle, as these devices are used exclusively in operating rooms.
Most importantly, however, was that the software needed to be able to detect diseased tissue in real-time as surgeons moved the detection probe over the patient’s body. In order to accomplish this, they needed a windows based tablet with the processing power necessary to avoid any delays whatsoever between the probe detecting diseased tissue and the alert sent to the surgeon.
 Solution
Solution
After struggling with the bulkiness and slow processing speed of a different tablet, the team at Intramedical was contacted by Cybernet. After demoing the CyberMed T10C medical tablet it became immediately clear that this was the device that they needed.
Not only was the CyberMed T10C a much more compact and sleek tablet, but it could easily be mounted on a tabletop mount or on a pole cart depending on each facility’s needs. It was IEC60601-1 certified and fanless - both important concerns for going to market quickly.
Most importantly, it was a Windows based tablet powered by an Intel quad core processor that would allow for real-time detection of diseased tissue without any lag or delay - the most critical specification that Intramedical needed to meet. Cybernet’s long product lifecycle also meant that Intramedical wouldn’t need to worry about getting their devices recertified every time a new model was released.
 Results
Results
The results since moving to Cybernet have been nothing short of spectacular. Intramedical Imaging was able to earn MDSAP (Medical Device Single Audit Program) certification, allowing them to distribute the Node Seeker 2000 in Australia, Brazil, Canada, Japan, and the USA. Despite a global customer base, Intramedical has reported nearly zero technical issues with the CyberMed T10C, and what few they have encountered were quickly resolved by Cybernet technical support team.
As the Node Seeker 2000 continues to be the leader in space, they continue to innovate. Intermedical Imaging is now looking to integrate their Node Seeker with the Davinci robotic surgery device and is looking for ways to provide the same real-time alerts to a surgeon working remotely, as they currently do to a surgeon physically in the operating room with the patient. When they make that leap forward, Cybernet will be right there with them.
We’ve been using the Cybernet tablets for 6 years now and we rarely have any issues with them. And when we do, we call your team, and within an hour the issue is rectified.
- Farhad Daghighian, CEO
INTRAMEDICAL IMAGING, LLC.