Device Maker Partners With Cybernet Creates Life Saving New Vital Sign

-
FLASHBACK TECHNOLOGIES
- Industry: Healthcare
- HQ: Louisville, CO
 Challenge
Challenge
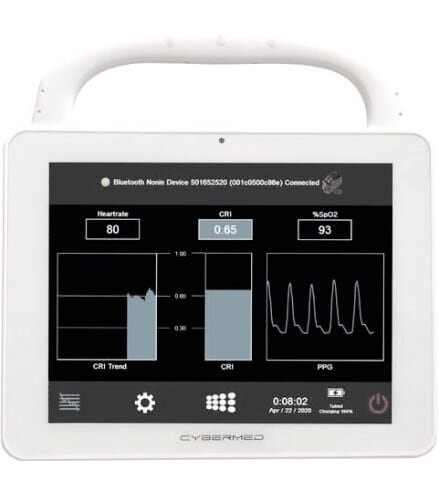
Flashback technologies had developed an algorithm called the Compensatory Reserve Index (CRI), which could more accurately predict when a patient was about to go into cardiovascular collapse due to traumatic blood loss. Often, patients can appear to be doing fine with traditional vital sign monitoring tools. However, they can quickly deteriorate without warning. By pairing their algorithm with a mobile device, they could give doctors and medics the tool they needed to better predict outcomes and save lives.
What flashback needed to accomplish this was a mobile device capable of running their software and integrating with their specialized oximeter. They required a touchscreen device with a clear display. But most importantly, because this device would be used in a clinical setting, they needed it to be medical grade. This meant having 60601-1 certification for near patient use, and the device also needed to be easily cleaned and disinfected between use.
 Solution
Solution
Having no interest in becoming a hardware manufacturer, Flashback Technologies set out to find a hardware partner that could provide an off-the-shelf solution that could run their software and check all the boxes. Finding a 60601-1 certified device was critical, but it also limited their options. The CyberMed T10C quickly emerged as a leading candidate. Its sturdy construction, 10” touch screen, and Windows operating system allowed it to run Flashback’s software easily. Beyond that, the CyberMed T10C had an IP65 sealed front bezel, allowing it to be sprayed down and cleaned between uses.
Equally important, Flashback needed a partner that could image their software and ship units directly to end users, taking Flashback completely out of the logistics business. This is a service that Cybernet offers to all of its device partners.
Another feature that wasn’t an initial consideration was the full sized ports available on the CyberMed T10C tablet. Flashback had initially been been using a Bluetooth oximeter, but the feedback they got from the field indicated that end users preferred a connected device. Cybernet was able to meet that demand without any further customization needed.
 Results
Results
Since first developing their software, Flashback has transitioned from a previous tablet model, the T10, to the newest version, the T10C. The consistency between the two devices has helped them navigate the FDA approval process, allowing them to commercialize their device for use in both a military setting - what it was originally designed for - and civilian environments.
In the civilian setting, end users report that they like the screen size, touch screen interface, and the desktop docking station. On the military side, having a device where all connectivity options - from ethernet to Bluetooth - could be completely disabled was a must have for use in the field. Overall, the feedback has been positive, with a few issues that Flashback has been able to work directly with Cybernet’s engineering team to fix.
Flashback Technologies is now in the process of going through the regulatory clearances to expand their indications for use on several different fronts and has been using the CyberMed T10C for all of that.
[Moving logistics operations] was huge. Cybernet was able to package everything together in one box and ship directly to our end customer. It makes my job a lot easier if we don’t have to manage an additional vendor.
- Grant Guenther, COO
FLASHBACK TECHNOLOGIES