Pioneering TeleHealth Device Maker Partners with Cybernet

-
MEDPOD, INC
- Industry: Healthcare
- Product: CyberMed PX22
- HQ: Melville, NY
 Challenge
Challenge
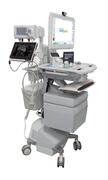
Medpod has long been an advocate of portable healthcare. The initial medpod devices were literal self contained pods that could be found in pharmacies and healthcare facilities. As technology has advanced, however, the team at Medpod decided to develop 3 different mobile telemedicine solutions. The first two would be placed on medical carts, one that was battery powered, and the other that was a non-powered cart. The third solution would be a portable telemedicine unit that could fold up into a suitcase when not in use.
When designing their carts, they decided that the most efficient way to navigate FDA approval would be to source each component for their final solution from vendors that already had medical grade certifications. Every part of the solution, from the cart and peripherals, and the monitor needed to be medical grade and safe to use near patients. Needless to say this created its own set of challenges. Naturally, when their search for a medical monitor for their cart began, they knew certain certifications such as UL60601-1 would be essential.
Finally, because portability was a primary function of these carts, whichever monitor they would decide to use would have to carry a small footprint. Something with enough screen space to allow for easy software navigation without overcrowding the cart that needed to also house other peripherals such as an ultrasound, scale, portable X-ray, and much, much more, would be preferred.
 Solution
Solution
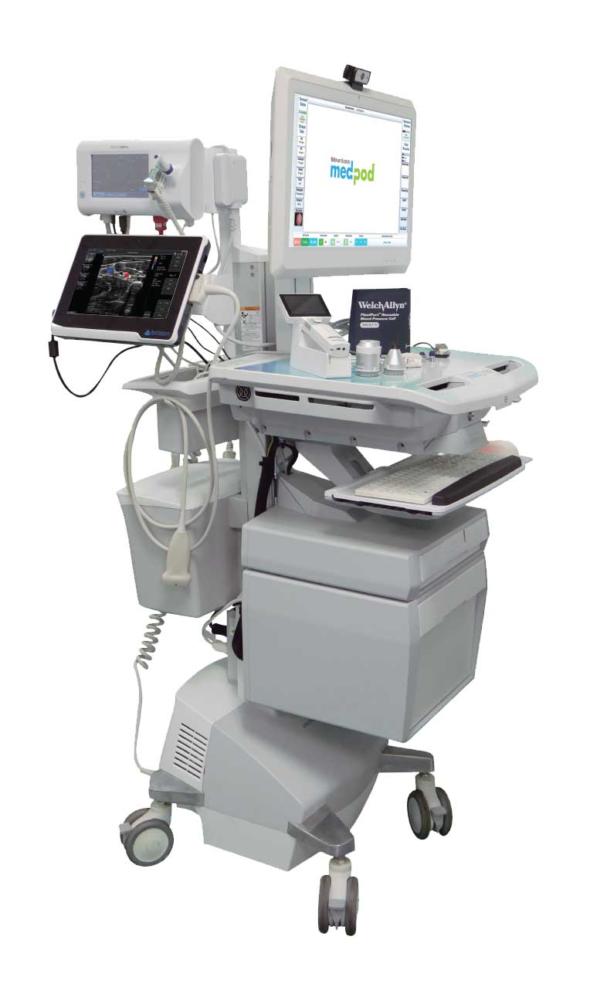
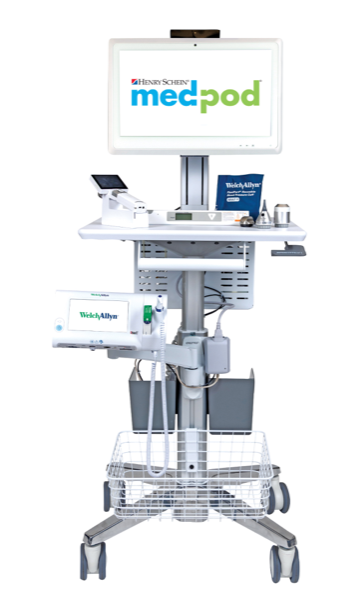
Dedicated to delivering only the best product to their customers, the team at Medpod tested and researched a slew of manufacturers and medical monitors before finally coming across the CyberMed PX22 Medical Monitor to pair with their powered cart solution.
Before any testing was done, the team was relieved to see that Cybernet’s solution met all of the medical grade requirements needed for a monitor that would be outfitted onto their carts. Firstly, the monitor carried global UL/cUL 60601-1 and IEC 60601-1-2 certifications, meaning they were rigorously tested and proven to be safe for near-patient use. Furthermore, having an IP65 certified front bezel meant the medical monitor could be regularly disinfected without worry over damaging the device’s internal components.
Aside from certifications, the Medpod team also observed in their testing that the 22 inch model of the CyberMed unit fit seamlessly onto their cart solutions among the multiple other devices held on said cart. The monitor’s moderate screen size in addition to its 75/100mm VESA mount compatibility meant it could be effortlessly attached to the Ergotron carts the team was already using without hassle.
Ultimately, the team had decided to move forward with Cybernet’s monitor solution, knowing that doing so would maintain their product’s FDA approval while also not compromising on the quality they promised to deliver to their customers.
 Results
Results
After deploying their telemedicine carts outfitted with the Cybernet unit, Medpod has continued to see growing interest in their telehealth products over the last couple of years. The team has received consistently positive feedback from their customers who have deployed their solutions in many practices ranging from physician offices and urgent care centers to hospitals and acute care facilities.
After their striking success with Cybernet’s PX22, the Medpod team is now considering using the CyberMed XB22 hot swap battery powered medical monitor for their non-powered cart solution. Because the XB22 carries all of the same medical grade certifications and has the same footprint, the team feels it would be a great fit. They are also considering Cybernet units for their suitcase solution, which currently uses a consumer grade tablet. As remote care becomes more and more the norm, the team is excited to further optimize their telemedicine solutions with devices like Cybernet’s that are safer, more lightweight, and carry a less space-consuming footprint.
It’s been fantastic. We’ve had local news stories where our customers are essentially running commercials for us since they’re so excited about our products and can’t stop raving about them.
- Lou Lavino, Senior Director, Implementation
MEDPOD, INC